Buy 2G-TZ 30mg Peptide: The Advanced Dual Receptor Agonist
Elevating Metabolic Research with the 2G-TZ 30mg Peptide
As the global scientific community continues to map the profound complexities of cellular metabolism, energy homeostasis, and endocrine signaling cascades, the demand for highly stable, multi-receptor agonists has exponentially increased. Enter the 2G-TZ 30mg Peptide, an industry-leading synthetic analog designed specifically to support rigorous, high-level in-vitro laboratory analysis, pancreatic beta-cell modeling, and scalable cellular screening.
This highly stable, lyophilized powder represents the absolute cutting edge of “second-generation” metabolic peptides. While much of the past decade’s research focused exclusively on single-pathway Glucagon-Like Peptide-1 (GLP-1) analogs, the 2G-TZ 30mg Peptide allows independent researchers and testing facilities to study the profound synergistic effects of dual-receptor agonism.
What truly sets this specific listing apart for the advanced laboratory is its versatile, mid-range capacity. By providing a precisely measured 30mg dosage in a sterile, vacuum-sealed glass vial, the 2G-TZ 30mg Peptide offers the perfect balance between extended assay capacity and targeted experimental control. This 30mg yield allows laboratories to standardize multiple runs of complex cellular assays from a single, unified source vial, effectively eliminating the risk of batch-to-batch molecular variation that can corrupt analytical data during medium-duration experiments.
The Biochemical Engineering of the 2G-TZ 30mg Peptide
To truly understand the profound analytical and research value of the 2G-TZ 30mg Peptide, scientists must first examine its brilliant structural engineering. The molecule is fundamentally a synthetic, 39-amino-acid peptide designed to simultaneously mimic two naturally occurring incretin hormones: Gastric Inhibitory Polypeptide (GIP) and Glucagon-Like Peptide-1 (GLP-1).
In natural biological systems, these endogenous incretin hormones are secreted by the intestines in response to nutrient intake. They rapidly bind to receptors on the pancreas to stimulate insulin secretion, while also interacting with the central nervous system to regulate energy expenditure. However, native human GIP and GLP-1 are notoriously unstable in laboratory environments. They possess extremely short half-lives (often just a few minutes) due to rapid enzymatic degradation by dipeptidyl peptidase-4 (DPP-4). This makes native incretins practically useless for extended in-vitro assays, as they break down before researchers can gather meaningful, long-term observational data.
The 2G-TZ 30mg Peptide circumvents this biological limitation through highly advanced molecular engineering. First, specific amino acid substitutions (particularly the inclusion of aminoisobutyric acid) are made at the enzymatic cleavage sites, effectively rendering the peptide completely invisible to the destructive DPP-4 enzyme.
Second, and most importantly, the peptide chain is intricately modified through the attachment of a C20 fatty diacid moiety via a highly specific hydrophilic linker. In an in-vitro environment, this specific molecular acylation allows the 2G-TZ 30mg Peptide to bind strongly, yet non-covalently, to blood albumin present in the culture medium. This drastically extends its half-life, providing researchers with an incredibly stable, sustained dual-receptor activation that native hormones and earlier first-generation peptides simply cannot achieve.
Synergistic Mechanisms: The Power of Dual Agonism
The clinical and analytical value of the 2G-TZ 30mg Peptide as a primary research tool lies entirely in its nature as a “Twincretin”—a highly potent, non-selective agonist of both the GIP and GLP-1 receptors. Unlike older synthetic peptides that target a single, isolated biological pathway, this compound interacts robustly with two distinct incretin systems simultaneously, allowing laboratories to study a vast array of amplified downstream cellular responses.
When introduced to complex cell cultures, the 2G-TZ 30mg Peptide exerts its effects by binding to specific G-protein-coupled receptors (GPCRs), subsequently activating the adenylate cyclase pathway and dramatically elevating intracellular levels of cyclic AMP (cAMP). This secondary messenger system is what triggers the specific biological cascades across the different cellular structures. Interestingly, this specific molecule exhibits an imbalanced binding affinity, heavily favoring the GIP receptor over the GLP-1 receptor, which opens unique avenues for cellular modeling.
The GLP-1 Receptor Pathway
The first major function of the 2G-TZ 30mg Peptide is its aggressive agonism of the GLP-1 receptor. GLP-1 receptors are heavily expressed on the surface of pancreatic beta cells—the highly specialized cells primarily responsible for producing and secreting insulin.
When researchers apply this peptide to isolated pancreatic cell cultures, the massive up-regulation of intracellular cAMP leads to a highly specific, glucose-dependent release of insulin stored in intracellular vesicles. In advanced in-vitro assays, laboratories utilize the 2G-TZ 30mg Peptide to study the exact stoichiometry and kinetics of insulin transcription and secretion. Furthermore, prolonged exposure to the GLP-1 agonism provided by this peptide has been observed to promote the actual proliferation and survival of beta cells, preventing cellular apoptosis (programmed cell death) when the culture is subjected to extreme oxidative stress or toxic lipotoxicity.
The GIP Receptor Pathway
While the GLP-1 pathway handles insulin secretion, the profound secondary agonism of the GIP receptor is what makes the 2G-TZ 30mg Peptide a revolutionary analytical tool. Historically, GIP was largely ignored by researchers because its insulinotropic effects are severely blunted in environments mirroring severe metabolic dysfunction. However, modern biochemistry has revealed that GIP plays a massive, unprecedented role in lipid metabolism and central energy homeostasis.
The 2G-TZ 30mg Peptide binds to the GIP receptor with an affinity comparable to native GIP itself. When introduced to specialized adipocyte (fat cell) cultures, the GIP agonism drastically alters how the cells handle lipids. It promotes the rapid clearance of triglycerides, increases insulin sensitivity within the fat cell itself, and modulates the secretion of pro-inflammatory adipokines.
When both the GIP and GLP-1 pathways are activated simultaneously within a single complex cell model, the resulting biological synergy is profound. The GIP agonism essentially “unlocks” the cellular environment, buffering the cells against inflammatory stress and allowing the GLP-1 agonism to operate at maximum efficiency. This synergistic “Twincretin” effect is why the compound is the premier choice for laboratories studying complex metabolic syndromes, advanced cellular lipotoxicity, and massive-scale energy homeostasis.
Verifiable Science Supporting the 2G-TZ 30mg Peptide
The complex biochemical mechanisms, acylated structural modifications, and profound dual-receptor agonism of twincretin analogs are extensively documented in modern, peer-reviewed scientific literature. Researchers investigating the fundamental properties of these advanced amino acid chains can find thousands of published studies detailing their physiological effects on isolated cell lines, pancreatic cultures, and highly complex metabolic animal models.
For highly authoritative, peer-reviewed data regarding the exact receptor-binding profiles, structural mapping, and vast physiological reach of GIP/GLP-1 dual agonists, researchers are highly encouraged to review extensive physiological studies via the National Center for Biotechnology Information (NCBI). Accessing this foundational research provides a rock-solid, verifiable scientific baseline for laboratories planning to utilize the 2G-TZ 30mg Peptide in their own novel analytical and metabolic experiments.
Ideal In-Vitro Applications for the 2G-TZ 30mg Peptide
Because of its unparalleled dual-pathway approach to the incretin system and its highly versatile mid-range vial capacity, the 2G-TZ 30mg Peptide is incredibly adaptable and can be deployed in a vast variety of scalable in-vitro assays. Laboratories purchasing this advanced compound frequently utilize it for the following primary research models:
-
Scalable Beta-Cell Assays: Utilizing the 30mg supply to standardize multiple isolated pancreatic beta-cell cultures, measuring the exact rate of glucose-dependent insulin secretion under the influence of dual-agonism, providing a perfect balance of volume for week-long assay blocks.
-
Adipocyte Lipolysis and Lipid Clearance Models: Applying the 2G-TZ 30mg Peptide to fat cell lines to map the exact GIP-receptor-mediated pathways responsible for clearing triglycerides, tracking the intracellular down-regulation of lipogenic enzymes over extended incubation periods.
-
Receptor Binding Affinity and Competitive Assays: Applying the compound alongside known selective GIP or GLP-1 antagonists (such as exendin-9,39) to map competitive binding kinetics, calculating precise IC50 (half-maximal inhibitory concentration) values to isolate the specific downstream effects of each receptor pathway.
-
Intracellular cAMP Quantification: Extracting cellular material from peptide-treated cultures to perform advanced assays, measuring the accumulation of cAMP using FRET sensors or ELISA to determine the exact magnitude of G-protein coupled receptor activation.
Independent Laboratory Testing and Purity Standards for the 2G-TZ 30mg Peptide
At Orbitrex, we intrinsically understand that your advanced analytical research data is only as accurate, reproducible, and strictly reliable as the raw materials you test. If a complex synthetic peptide contains micro-impurities, excess salts, incomplete acylation, or degraded amino acid chains, it will instantly introduce unpredictable variables into your research, effectively rendering your expensive analytical data completely useless.
This is precisely why we guarantee that every single batch of our 2G-TZ 30mg Peptide undergoes the most rigorous, independent third-party testing available before it is ever cleared for distribution to the scientific community.
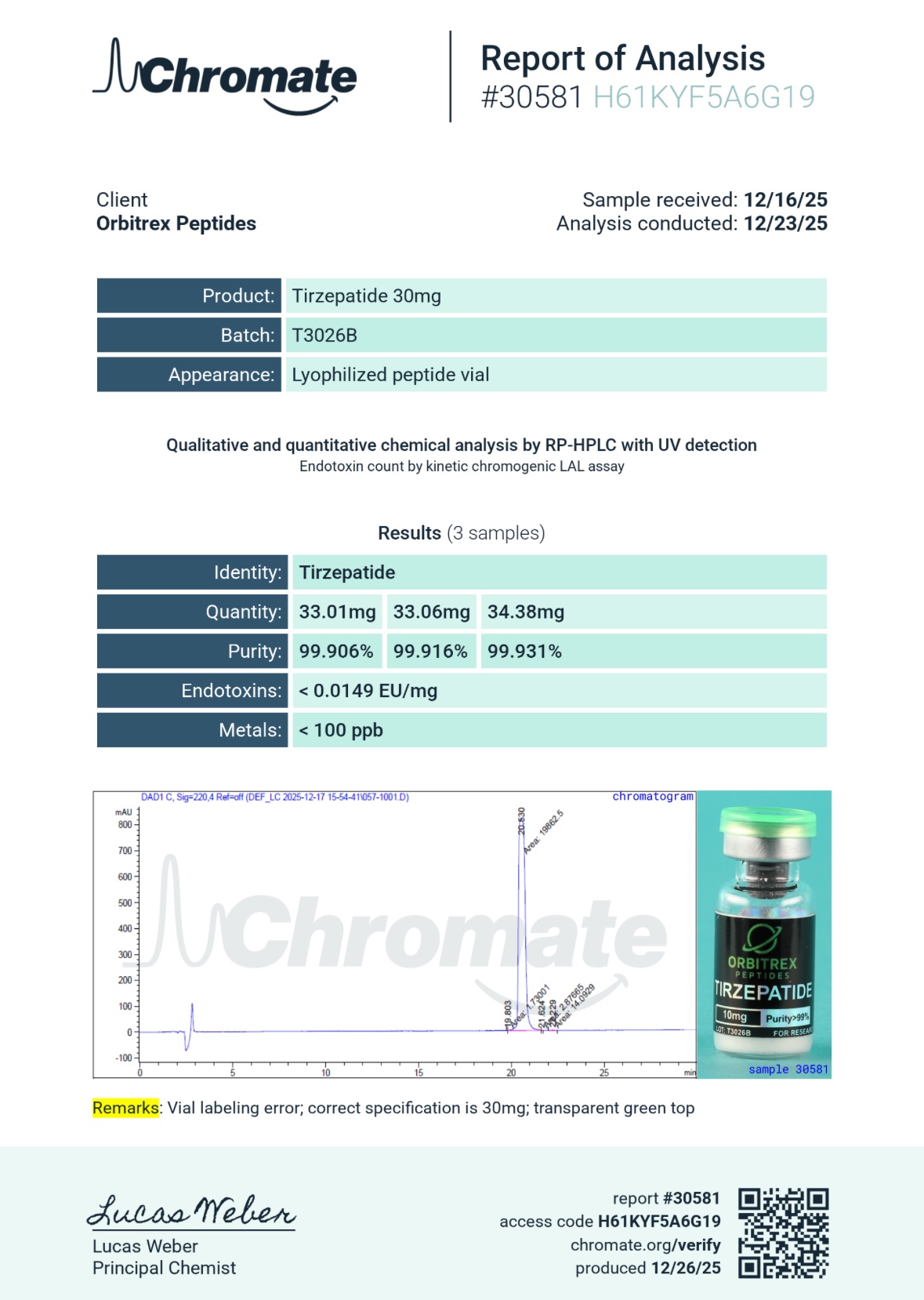
Our primary testing methodologies include High-Performance Liquid Chromatography (HPLC) and Liquid Chromatography-Mass Spectrometry (LC-MS). The HPLC analysis ensures that the molecular weight and retention time of the powder perfectly match the exact profile of pure dual-agonist incretins, successfully separating out any potential biological contaminants, leftover manufacturing solvents, or truncated peptide fragments.
The Mass Spectrometry confirms the exact 39-amino-acid sequence of the peptide and specifically verifies the successful attachment of the critical C20 fatty diacid chain. Manufacturing an acylated peptide of this length requires immense biochemical precision; our LC-MS testing guarantees there are no missing, swapped, or malformed links in the molecular structure.
We mandate strictly >99% purity for the 2G-TZ 30mg Peptide, ensuring your laboratory receives uncompromised materials entirely free from binders, heavy metals, synthetic impurities, or leftover manufacturing byproducts. When you source your complex metabolic materials from Orbitrex, you are sourcing absolute, verifiable reliability.
Preparation and Handling of the 2G-TZ 30mg Peptide
Due to the highly fragile molecular structure of synthetic amino acid chains, the proper preparation, reconstitution, and storage of the 2G-TZ 30mg Peptide are absolutely critical to maintaining its analytical efficacy and preventing rapid degradation prior to your planned experiments.
The product is shipped as a lyophilized (freeze-dried) solid powder puck inside a sterile, vacuum-sealed glass vial. Lyophilization removes the water from the peptide solution under extremely low temperature and pressure, which stabilizes the fragile molecular bonds and allows the 2G-TZ 30mg Peptide to survive the shipping process without rapid degradation or structural collapse.
Reconstitution Protocols
To prepare the 2G-TZ 30mg Peptide for in-vitro application, the powder must be carefully reconstituted into a liquid solution. Because this is a 30mg vial, researchers must carefully calculate the required volume of diluent to achieve their desired mg/mL concentration (e.g., adding 3mL of diluent will yield a 10mg/mL solution). It is highly recommended to use sterile bacteriostatic water (water containing 0.9% benzyl alcohol) or sterile saline, depending entirely on the specific chemical requirements and osmolality of your specific cell culture.
When introducing the diluent into the vial containing the 2G-TZ 30mg Peptide, standard sterile laboratory protocols must be strictly observed:
-
Swab the rubber stopper of both the peptide vial and the diluent vial with 70% isopropyl alcohol to ensure complete sterility.
-
Using a sterile syringe, draw the exact desired volume of diluent.
-
Inject the diluent extremely slowly into the peptide vial. Crucial: Direct the stream of the liquid against the inner glass wall of the vial rather than shooting it directly into the lyophilized powder. The force of a direct, high-pressure liquid stream can physically shear the delicate peptide bonds of the 2G-TZ 30mg Peptide, irreparably damaging the molecules before they are ever utilized in your assays.
-
Do not shake the vial under any circumstances. Vigorous shaking will destroy the amino acid sequences and disrupt the acylated structures. Gently swirl or roll the vial between your fingers until the powder has completely dissolved into a perfectly clear, uniform solution.
Storage Guidelines for the 2G-TZ 30mg Peptide
Maintaining the molecular integrity of the 2G-TZ 30mg Peptide post-reconstitution is paramount to the success, accuracy, and reproducibility of your scalable research blocks.
Unreconstituted (Lyophilized Powder): If you are not utilizing the peptide immediately upon delivery, the dry powder should be stored away from direct sunlight in a completely dry, temperature-controlled environment. For short-term storage (up to 30 days), standard room temperature is acceptable, though refrigeration is always preferred. For long-term storage (up to 24 months), the lyophilized vials must be kept in a dedicated laboratory freezer at -20°C.
Reconstituted (Liquid Solution): Once bacteriostatic water or saline has been introduced to the 2G-TZ 30mg Peptide, the structural degradation clock begins ticking immediately. The reconstituted vial must be immediately refrigerated at 2°C to 8°C (36°F to 46°F). Even when kept at precise, temperature-controlled refrigerated settings, the mixed solution must be utilized in your laboratory assays within 20 to 30 days. Beyond this tight window, the delicate peptide chains will begin to naturally degrade, losing their biological potency, breaking down into unusable fragments, and inevitably skewing your analytical data.
Disclaimer: All products listed by Orbitrex, including the 2G-TZ 30mg Peptide, are sold strictly for in-vitro laboratory research and analytical purposes only. They are not intended for human consumption, ingestion, diagnostic, therapeutic, or agricultural use. Our products are not FDA-approved for human use. Any communications implying human use, clinical trials on unapproved human subjects, or bodily injection will result in the immediate cancellation of your account and permanent restriction from our supply chain. All researchers must adhere to their local institutional review board (IRB) guidelines when handling these research materials.