Buy 3G-RT 10mg Peptide: The Advanced Triple Receptor Agonist
Elevating Precision Metabolic Research with the 3G-RT 10mg Peptide
As the global scientific community continues to meticulously map the profound, cascading complexities of cellular metabolism, hepatic energy homeostasis, and intricate endocrine signaling, the demand for highly stable, multi-receptor agonists has exponentially increased. Enter the 3G-RT 10mg Peptide, an industry-leading synthetic analogue designed specifically to support rigorous, high-level in-vitro laboratory analysis, targeted hepatic cell modeling, and precision dose-escalation screening.
This highly stable, lyophilized powder represents the absolute cutting edge of “third-generation” metabolic peptides. While much of the past decade’s research focused first on single-pathway Glucagon-Like Peptide-1 (GLP-1) analogues, and subsequently on dual-pathway “twincretins,” the 3G-RT 10mg Peptide represents a massive biochemical leap forward. It allows independent researchers and advanced testing facilities to study the profound, unprecedented synergistic effects of a triple-receptor agonist in a highly controlled, synthetic environment.
What truly sets this specific listing apart for the advanced laboratory is its identity as the precision baseline unit. By providing a specifically measured 10mg dosage in a sterile, vacuum-sealed glass vial, the 3G-RT 10mg Peptide offers the perfect, standardized volume for laboratories conducting isolated control experiments, targeted pilot studies, or establishing exact comparative baselines against first-generation compounds. This 10mg yield is critical for initial assay design; it allows researchers to rigorously test complex triple-agonist dose-escalation hypotheses on specialized cell lines without committing to the massive volumes required for industrial-scale screening. This ensures highly efficient use of expensive laboratory resources, minimizes chemical waste, and maintains absolute structural purity for localized, individual baseline experiments.
The Biochemical Engineering of the 3G-RT 10mg Peptide
To truly understand the profound analytical and research value of the 3G-RT 10mg Peptide, scientists must first deeply examine its brilliant, unprecedented structural engineering. The molecule is fundamentally a synthetic, 39-amino-acid peptide designed to simultaneously mimic three distinct, naturally occurring metabolic hormones: Glucagon-Like Peptide-1 (GLP-1), Gastric Inhibitory Polypeptide (GIP), and native Glucagon (GCG).
In natural biological systems, these endogenous hormones operate on entirely different, sometimes directly opposing, physiological axes. GLP-1 and GIP are incretins secreted by the intestinal tract to stimulate insulin secretion, preserve pancreatic beta-cells, and regulate nutrient intake. Glucagon, conversely, is secreted by the alpha cells of the pancreas to raise blood sugar and rapidly mobilize stored cellular energy. Achieving simultaneous agonism of all three of these receptors using a single, unified molecule is considered an absolute masterpiece of modern synthetic biochemistry.
However, native human hormones are notoriously unstable in laboratory environments. They possess extremely short half-lives (often just a few minutes) due to rapid, aggressive enzymatic degradation by dipeptidyl peptidase-4 (DPP-4) and other circulating proteases.
The 3G-RT 10mg Peptide circumvents these severe biological limitations through highly advanced molecular engineering. First, specific amino acid substitutions (particularly the inclusion of aminoisobutyric acid, or Aib, at position 2) are made directly at the primary enzymatic cleavage sites. This crucial substitution provides profound steric hindrance, effectively rendering the entire 39-chain peptide completely invisible to the destructive DPP-4 enzyme, preserving the integrity of the molecule.
Second, the peptide chain is intricately modified through the precise attachment of a C20 fatty diacid moiety via a highly specific, hydrophilic chemical linker. In an in-vitro cell culture environment, this specific molecular acylation allows the 3G-RT 10mg Peptide to bind strongly, yet non-covalently, to blood albumin present in the culture medium. This drastically extends its functional half-life, providing researchers with an incredibly stable, sustained triple-receptor activation that native hormones simply cannot achieve in a standard petri dish.
Synergistic Mechanisms: The Power of Triple Agonism
The clinical and analytical value of the 3G-RT 10mg Peptide as a primary research tool lies entirely in its nature as a “Tri-agonist”—a highly potent, non-selective activator of the GLP-1, GIP, and GCGR receptors. Unlike older synthetic peptides that target one or two isolated biological pathways, this compound interacts robustly with three distinct metabolic systems simultaneously, allowing laboratories to study a vast array of amplified, interconnected downstream cellular responses from a single precision dose.
When introduced to complex, multi-tissue cell cultures, the 3G-RT 10mg Peptide exerts its effects by binding to specific G-protein-coupled receptors (GPCRs), subsequently activating the adenylate cyclase pathway and dramatically elevating intracellular levels of cyclic AMP (cAMP) across multiple distinct cell types.
The GLP-1 Receptor Pathway: Beta-Cell Activation and Preservation
The first major foundational function of the 3G-RT 10mg Peptide is its aggressive agonism of the GLP-1 receptor. GLP-1 receptors are heavily expressed on the surface of pancreatic beta cells—the highly specialized endocrine cells primarily responsible for producing, storing, and secreting insulin.
When researchers apply this highly stabilized 10mg peptide to isolated pancreatic cell cultures (such as INS-1 or MIN6 cell lines), the massive up-regulation of intracellular cAMP leads to a highly specific, glucose-dependent release of insulin stored in intracellular vesicles via the targeted activation of Protein Kinase A (PKA). In advanced in-vitro assays, laboratories utilize the 3G-RT 10mg Peptide to study the exact stoichiometry and kinetics of insulin transcription. Furthermore, prolonged exposure to the GLP-1 agonism provided by this peptide promotes the actual proliferation of beta cells. It directly prevents cellular apoptosis (programmed cell death) by up-regulating vital anti-apoptotic proteins (like Bcl-2) when the culture is subjected to extreme oxidative stress, endoplasmic reticulum (ER) stress, or toxic glucotoxicity.
The GIP Receptor Pathway: Adipocyte Modulation and Anti-Inflammatory Buffering
While the GLP-1 pathway handles critical insulin secretion, the profound secondary agonism of the GIP receptor allows the 3G-RT 10mg Peptide to act heavily on localized lipid tissues. Modern biochemistry has revealed that GIP plays a massive, unprecedented role in lipid metabolism and cellular energy homeostasis, acting as the perfect bridge between insulin secretion and energy expenditure.
The 3G-RT 10mg Peptide binds to the GIP receptor with incredibly high affinity. When introduced to specialized adipocyte (fat cell) cultures (such as differentiated 3T3-L1 cell lines), the GIP agonism drastically alters how the cells handle dense, stored lipids. It promotes the rapid intracellular clearance of triglycerides, radically increases insulin sensitivity within the fat cell itself, and powerfully modulates the secretion of pro-inflammatory adipokines. This creates a highly buffered, anti-inflammatory cellular environment that primes the culture for the third, most aggressive stage of the peptide’s tri-agonist mechanism.
The Glucagon Receptor (GCGR) Pathway: Hepatic Energy Expenditure
The absolute defining characteristic that separates the 3G-RT 10mg Peptide from previous generations of research chemicals is its potent, controlled agonism of the Glucagon Receptor (GCGR). This is the biochemical game-changer for modern metabolic research.
Glucagon receptors are heavily concentrated in hepatocytes (liver cells). In isolated nature, agonizing the glucagon receptor would normally cause the liver cell to aggressively dump glucose into the bloodstream (glycogenolysis), which would trigger massive metabolic instability. However, within the brilliantly engineered framework of this triple-agonist, the glucose-dumping effect of GCGR is completely neutralized and overpowered by the insulin-stimulating effects of the GLP-1 and GIP pathways.
So, what is the biological purpose of activating the glucagon receptor within this specific complex? Massive, unprecedented mitochondrial energy expenditure and direct hepatic lipid clearance.
When laboratories introduce the 3G-RT 10mg Peptide to human hepatocyte cell cultures modeled to simulate severe Non-Alcoholic Fatty Liver Disease (NAFLD) or cellular steatosis, the GCGR activation triggers extreme intracellular lipid oxidation. The hepatocytes are forced to rapidly burn through their stored toxic lipid droplets for immediate energy. Furthermore, GCGR activation powerfully up-regulates mitochondrial respiration, drastically increasing the basal metabolic rate of the cell itself.
The resulting biological synergy is profound. The GCGR pathway aggressively shreds the toxic liver fat and ramps up mitochondrial oxygen consumption, while the GLP-1 and GIP pathways ensure the cellular environment remains highly sensitive to insulin. This unique, three-pronged mechanism is exactly why the compound is the premier choice for laboratories studying complex metabolic syndromes, advanced hepatic lipotoxicity, and massive-scale cellular energy expenditure during highly targeted, single-run analytical blocks.
Verifiable Science Supporting the 3G-RT 10mg Peptide
The complex biochemical mechanisms, acylated structural modifications, and profound triple-receptor agonism of third-generation incretin analogues are extensively documented in modern, heavily peer-reviewed scientific literature. Researchers investigating the fundamental properties of these highly advanced 39-amino-acid chains can find thousands of published studies detailing their physiological effects on isolated hepatic cell lines, pancreatic cultures, and highly complex metabolic animal models.
For highly authoritative, peer-reviewed data regarding the exact receptor-binding profiles, structural mapping, and vast physiological reach of GLP-1/GIP/GCGR triple agonists, researchers are highly encouraged to review extensive physiological studies via the National Center for Biotechnology Information (NCBI). Accessing this foundational research provides a rock-solid, verifiable scientific baseline for laboratories planning to utilize the 3G-RT 10mg Peptide in their own novel analytical, metabolic, and targeted comparative baseline experiments.
Ideal In-Vitro Applications for the 3G-RT 10mg Peptide
Because of its unparalleled triple-pathway approach to the incretin and glucagon systems, combined with its highly precise, industry-standard 10mg vial capacity, the 3G-RT 10mg Peptide is incredibly adaptable and designed exclusively for targeted, precision in-vitro assays. Laboratories purchasing this advanced compound frequently utilize it for the following primary research models:
-
Targeted Pilot Studies and Dose-Escalation Baselines: Utilizing the precise 10mg supply to establish an exact baseline cellular response for triple-agonism. Researchers run this formulation across a vast gradient of microgram-per-milliliter concentrations to pinpoint the exact dosage required to achieve maximal hepatic lipid clearance before committing to massive, expensive master-batches for larger assays.
-
Isolated Comparative Efficacy Assays: Applying the 3G-RT 10mg Peptide in direct, side-by-side comparison with dual-agonists (like Tirzepatide analogues) or single-agonists (like Semaglutide analogues). This allows laboratories to perfectly isolate and quantify the exact biological advantage provided strictly by the addition of the Glucagon Receptor (GCGR) pathway in a perfectly controlled, short-term assay.
-
Mitochondrial Respiration and OCR Quantification: Extracting cellular material from targeted, peptide-treated cultures to perform highly advanced metabolic assays. Laboratories utilize advanced cellular machinery (like Seahorse XF Analyzers) to track the acute, rapid up-regulation of the mitochondrial oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) driven specifically by the glucagon pathway.
-
Receptor Binding Affinity and Triple-Competitive Kinetics: Applying the compound alongside known selective GLP-1, GIP, and GCGR antagonists in targeted, single-run batches. This allows researchers to map incredibly complex competitive binding kinetics, calculating precise IC50 values to isolate the specific downstream effects of each of the three receptor pathways in real-time.
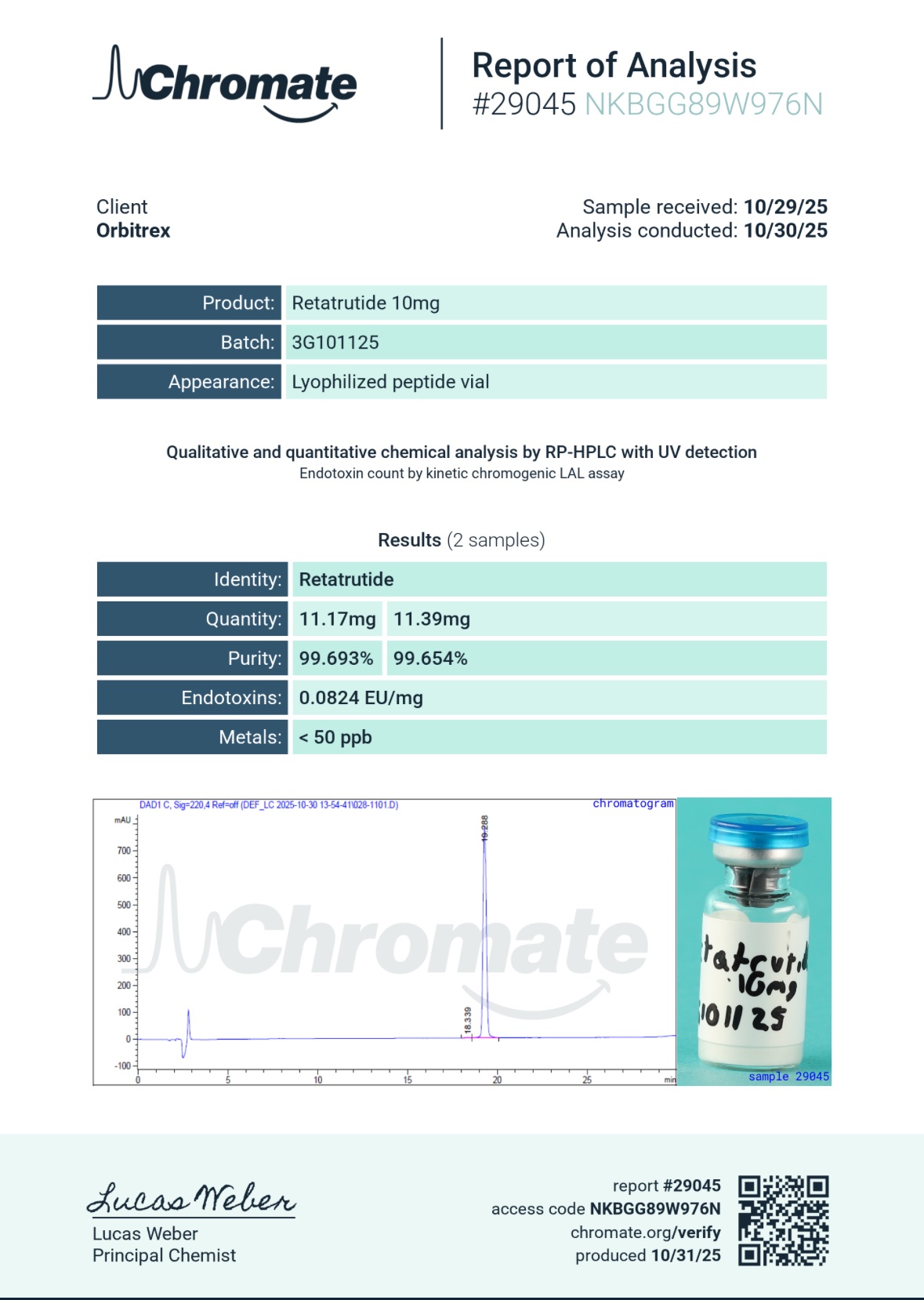
Independent Laboratory Testing and Purity Standards for the 3G-RT 10mg Peptide
At Orbitrex, we intrinsically understand that your advanced analytical research data is only as accurate, reproducible, and strictly reliable as the raw materials you test. When conducting targeted pilot studies utilizing a 10mg precision vial, a single micro-impurity can skew the entire trajectory of an expensive, multi-stage research project. If a complex synthetic peptide contains micro-impurities, excess salts, incomplete acylation, or degraded amino acid chains, it will instantly introduce unpredictable variables into your research, effectively rendering your analytical data completely useless.
This is precisely why we guarantee that every single batch of our 3G-RT 10mg Peptide undergoes the most rigorous, independent third-party testing available before it is ever cleared for distribution to the scientific community.
Our primary testing methodologies include High-Performance Liquid Chromatography (HPLC) and Liquid Chromatography-Mass Spectrometry (LC-MS). The HPLC analysis ensures that the molecular weight and retention time of the powder perfectly match the exact profile of pure triple-agonist incretins, successfully separating out any potential biological contaminants, leftover manufacturing solvents, or truncated peptide fragments.
The Mass Spectrometry confirms the exact 39-amino-acid sequence of the peptide and specifically verifies the successful attachment of the critical C20 fatty diacid chain. Manufacturing an acylated triple-agonist peptide of this length requires immense biochemical precision; our LC-MS testing guarantees there are no missing, swapped, or malformed links in the molecular structure.
We mandate strictly >99% purity for the 3G-RT 10mg Peptide, ensuring your laboratory receives uncompromised materials entirely free from binders, heavy metals, synthetic impurities, or leftover manufacturing byproducts. When you source your complex metabolic materials from Orbitrex, you are sourcing absolute, verifiable reliability for your precision baseline assays.
Preparation and Handling of the 3G-RT 10mg Peptide
Due to the highly fragile molecular structure of synthetic amino acid chains, the proper preparation, reconstitution, and storage of the 3G-RT 10mg Peptide are absolutely critical to maintaining its analytical efficacy and preventing rapid degradation prior to your planned experiments.
The product is shipped as a lyophilized (freeze-dried) solid powder puck inside a sterile, vacuum-sealed glass vial. Lyophilization removes the water from the peptide solution under extremely low temperature and pressure, which stabilizes the fragile molecular bonds and allows the 3G-RT 10mg Peptide to survive the domestic shipping process without rapid degradation, spontaneous aggregation, or structural collapse.
Reconstitution Protocols
To prepare the 3G-RT 10mg Peptide for in-vitro application, the powder must be carefully reconstituted into a liquid solution. Because this is an industry-standard 10mg precision vial, researchers can easily calculate the required volume of diluent to achieve their desired mg/mL concentration (e.g., adding 1.0mL of diluent will yield an incredibly dense 10mg/mL solution, or 2.0mL for a highly workable 5mg/mL yield). It is highly recommended to use sterile bacteriostatic water (water containing 0.9% benzyl alcohol) or sterile saline, depending entirely on the specific chemical requirements, pH, and osmolality of your specific multi-well cell cultures.
When introducing the diluent into the vial containing the 3G-RT 10mg Peptide, standard sterile laboratory protocols must be strictly observed:
-
Swab the rubber stopper of both the peptide vial and the diluent vial with 70% isopropyl alcohol to ensure complete, uncompromised sterility.
-
Using a sterile syringe, draw the exact desired volume of diluent required for your targeted baseline yield.
-
Inject the diluent extremely slowly into the peptide vial. Crucial: Direct the stream of the liquid against the inner glass wall of the vial rather than shooting it directly into the lyophilized powder puck. The force of a direct, high-pressure liquid stream can physically shear the delicate 39-chain peptide bonds of the 3G-RT 10mg Peptide, irreparably damaging the molecules before they are ever utilized in your expensive assays.
-
Do not shake the vial under any circumstances. Vigorous shaking will destroy the amino acid sequences and violently disrupt the acylated structures. Gently swirl or roll the vial between your fingers until the powder puck has completely dissolved into a perfectly clear, uniform solution.
Storage Guidelines for the 3G-RT 10mg Peptide
Maintaining the molecular integrity of the 3G-RT 10mg Peptide post-reconstitution is paramount to the success, accuracy, and reproducibility of your highly targeted, comparative research blocks.
Unreconstituted (Lyophilized Powder): If you are not utilizing the peptide immediately upon delivery, the dry powder should be stored away from direct sunlight in a completely dry, temperature-controlled environment. For short-term storage (up to 30 days), standard room temperature is acceptable, though refrigeration is always strongly preferred. For long-term storage (up to 24 months), the lyophilized vials must be kept in a dedicated laboratory freezer at -20°C.
Reconstituted (Liquid Solution): Once bacteriostatic water or saline has been introduced to the 3G-RT 10mg Peptide, the structural degradation clock begins ticking immediately. Because this 10mg vial is designed to establish highly precise baseline measurements, storage protocols must be incredibly strict. The reconstituted vial must be immediately refrigerated at 2°C to 8°C (36°F to 46°F). Even when kept at precise, temperature-controlled refrigerated settings, the mixed solution must be utilized in your laboratory assays within 20 to 30 days. Beyond this tight window, the delicate 39-chain peptide will begin to naturally degrade, losing its biological potency, breaking down into unusable fragments, and inevitably skewing your vital analytical baseline data.
Disclaimer: All products listed by Orbitrex, including the 3G-RT 10mg Peptide, are sold strictly for in-vitro laboratory research and analytical purposes only. They are not intended for human consumption, ingestion, diagnostic, therapeutic, or agricultural use. Our products are not FDA-approved for human use. Any communications implying human use, clinical trials on unapproved human subjects, or bodily injection will result in the immediate cancellation of your account and permanent restriction from our supply chain. All researchers must adhere to their local institutional review board (IRB) guidelines when handling these research materials.